Why Ballistocardiography is the Leader of Next Generation Cardiac Sensors
Published: May 22, 2023
Casana’s smart toilet seat for heart health can capture clinical measurements like blood oxygenation, heart rate, with blood pressure in development. We’re asked all the time; how? The key lies inside our bumpers, or rather, our ballistocardiograms.
The Ballistocardiogram
The recoil of the human body each time the heart ejects blood into the arteries was discovered over 150 years ago. To observe this force of nature all you need to do is hold your breath and stand perfectly still on an old analog scale; the needle on the scale will oscillate with the force of your heartbeat, as the subtle force of blood pumping and pressure waves moving, creates a minuscule but measurable force.
Ballistocardiography (BCG) is a non-invasive method for monitoring this cardiovascular phenomenon. Although initially intended for in-clinic use, the BCG has recently gained renewed attention from medical professionals for its potential outside of clinical settings, including home monitoring. This shift towards new applications can be attributed to recent advancements in technology, such as sensor miniaturization. Modern BCG devices are compact, user-friendly, and noninvasive, which has reestablished clinical interest in BCG applications by the medical community. So why is it that the BCG is only now seeing a resurgence of popularity and development, and what can we expect for the future of this underutilized technology? To address these questions, we first have to take a step back and explore the historical context of the rise and fall of the BCG throughout the 1900s.
Origins: 1870s-1950s
Prior to the development of the echocardiograph (echo) in 1952, physicians struggled to quantify the mechanical forces of the heart (e.g. cardiac output). The recognition that cardiovascular forces have an effect on the recoil movements of the human body—the key to unlocking ballistocardiography (BCG)—was first studied as early as 1877 by J.W. Gordon. Although a BCG could show abnormalities in the presence of heart failure, several factors made reproducible results challenging. One study noted that during inspiration and expiration, a ballistocardiogram would record ballistic impulse rather than actual blood ejection or systolic output, thus giving a false impression of results.
Despite early efforts by a multitude of scientists, no real advances were made in the technique or application of BCG monitoring until Dr. Issac Starr (and associates) reported their original investigations in 1939. Starr and his associates constructed a “high frequency” bed and studied its physical properties along with those of the human body.
So how did early BCGs work? Patients would lay on a bed or couch that was mounted on a platform and could move freely in any direction. When the heart contracted and ejected blood into the aorta, the entire body would recoil slightly in the opposite direction. This recoil was transmitted to the platform, which was equipped with a pen that traced the movement on a moving chart. The recordings from this bed were then termed “ballistocardiographs”.

Figure 1—High-frequency ballistocardiogram, commercial model (1952)
Starr later developed formulas for estimating cardiac output and began to carry out extensive clinical studies. His reports of progress stimulated many other investigators to do the same, creating what may be called the BCG’s golden age of publications. One list compiled by the Federal Aviation Agency, published under joint auspices of the FAA and NASA, found over 1500 publications on ballistocardiography by 1965. However, this popularity did not last long.
The Decline: 1960s-1990s
After the introduction of the echocardiograph—a device capable of visualizing the activity of the heart through the use of high-frequency sound waves—the popularity of BCGs had a stark decline. A study on the resurgence of ballistocardiography by Laurent Giovangrandi et al. tracked BCG-related publications over a period of 100 years and found a significant downturn after the 1960s.
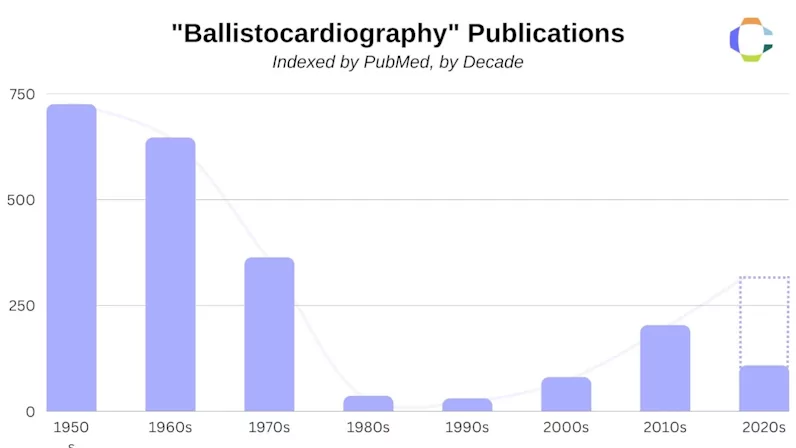
Figure 2—Ballistocardiography publications by decade
So why did the popularity of BCGs crash so ubiquitously after the introduction of the echocardiogram? One of the most influential reasons revolves around the minimal guidance available on how to analyze cardiac metrics with the BCGs of the time. When compared to the echocardiogram, which allowed physicians to see the mechanical activity of the heart as it beat in real-time, the BCG fell short and was quickly replaced over the span of a decade. At the time, there was still little clinical evidence that explained what BCGs actually measured and how physicians could use this data in a medical context.
According to R.S. Gubner et al., in regard to Starr’s longitudinal BCG, there was a general agreement amongst researchers that the early peak in a ballistocardiograph is related to the motion of the heart early in systole, while the main IJK complex (as seen below) is related to the ventricular ejection and aortic flow. For the later waves, however, clinicians were less sure what they represented. This factor in particular was one of the great barriers to the BCG’s adoption.
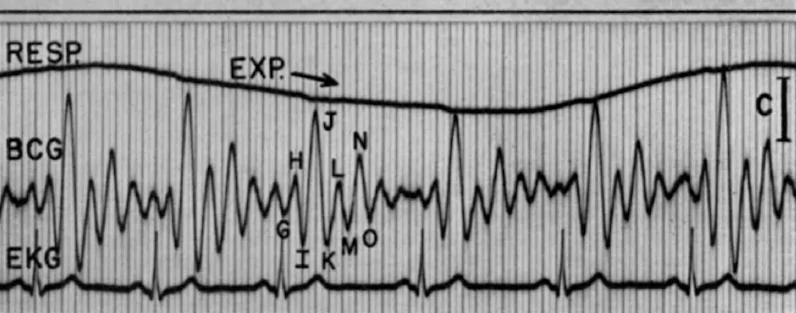
Figure 3—Results of a High-frequency ballistocardiograph (1952)
Finally, and perhaps most obvious, was the size of the device. Early BCGs were typically in the form of a bed, sometimes hanging, other times floating on a bed of mercury, making it extraordinarily costly for the average physician to acquire and use. The first factor in the revival of the BCG that addressed this problem was the development of sensor technology and miniaturization.
The Revival: 2000s-2020s
Since 2003, publications related to BCGs began to reappear within the scientific community. The reasons for this are multifaceted but can be distilled into two key factors: the miniaturization of sensors, and the development of novel technologies & signal-processing methods. Both of these changes facilitated the renewed usage of BCGs, as monitoring and analysis became more systematic and structured.
To put it simply, the newest iterations of the BCG sensors were small enough to be integrated into unique form factors aimed at addressing problems from home adherence to human error, while also being far more cost-effective than their historical counterparts. Modern BCGs can be as small as three to four inches in diameter, and as large as manufacturers may need to cover the required area of their device’s form factor.
Beyond the technological advancements making the revitalization of the BCG possible, the scientific community has also recently been motivated by the need to find better techniques for assessing the mechanical aspects of cardiovascular health outside of the clinic. The BCG is appealing in this regard due to its potential for improving the availability of clinically relevant information regarding cardiac output, cardiac contractility, and beat-by-beat left ventricular function from home. Compared to invasive, intrusive, or expensive alternatives, the BCG is currently a promising contender as the leader of the next generation of cardiac sensors.
Ballistocardiography in the 21st Century
Modern-day BCGs can be embedded into devices with a wide range of form factors, and often provide extra data points that can be used to determine overall cardiac health. A few of the modalities where BCGs have been implemented include smart bed sensors, smart scales, smart chairs, and even smart toilet seats. Two of the most alluring features of BCG sensors to modern researchers are their potential to measure cardiac metrics non-invasively, and how the sensors can be combined with other sensors such as SCGs, ECGs, photoplethysmograms (PPGs), etc.
Clinical Uses
While form factors vary, some of the most popular sensor combinations with the ballistocardiogram include, PPGs and ECGs or BCGs with SCGs. The former was recently studied for its potential to measure cuffless blood pressure, while the latter has been used to track cardiac activity analysis, respiratory monitoring, and sleep screening. One study, for example, developed a complete smart home health monitoring system through the combination of signals such as ECG, PPG, electromyography (EMG), BCG, and imaging photoplethysmography (iPPG).
In a review of more than 60 studies focusing on BCG utilization in both clinical and non-clinical settings, researchers found that heart rate was successfully recorded in 90% of the studies, while nearly half of the studies were able to discern respiratory rate. Other various metrics that could be determined by these devices included blood pressure, apneas, heart rate variation, breathing rate, and even left ventricular ejection time. Nearly all of the studies utilized a combination of sensors to achieve these results.
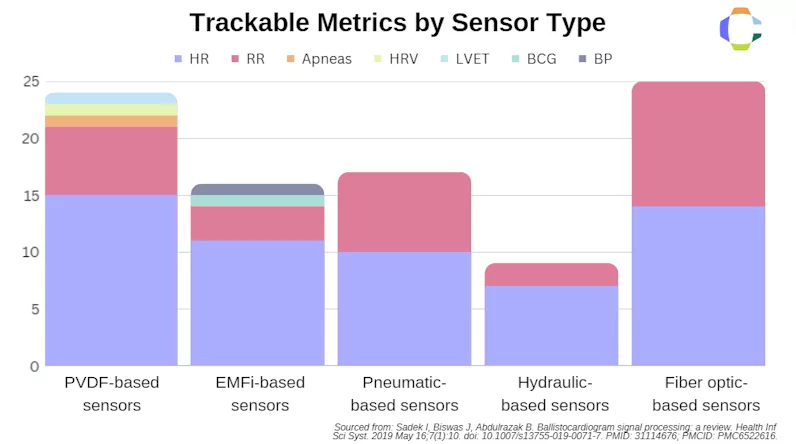
Figure 4—Chart of metrics tracked by sensor type as reviewed by Sadek et al.
The potential fusion of multiple sensors into one device is beneficial, as the health data can give clinicians a more comprehensive look at their patient’s overall health. Many businesses are considering sensor fusion within their own devices, yet few of these companies have added a ballistocardiogram. The reason for this lack of integration is that BCGs, despite their utility in providing monitoring insights, can be difficult to integrate into wearables or handheld devices. In layman’s terms, a BCG is essentially an extremely sensitive weight load cell. In order for it to work the BCG requires the user to place at least part of their body weight on the sensor. For the vast majority of wearables, this is impossible. In the context of device development, this means that researchers and innovators have to be far more creative in developing the form factor of their BCG monitoring solutions.
The Future of BCGs
The widespread use of ballistocardiograms in medical devices could influence the healthcare industry due to their unique form factor applications. Enhanced BCG technology presents exciting opportunities, particularly for in-home patient monitoring. BCGs offer a non-invasive, affordable method for tracking cardiovascular health outside traditional clinical environments. This development can be especially beneficial for patients with chronic conditions that require regular check-ins as well as for those with comorbidities who struggle to monitor multiple health metrics.
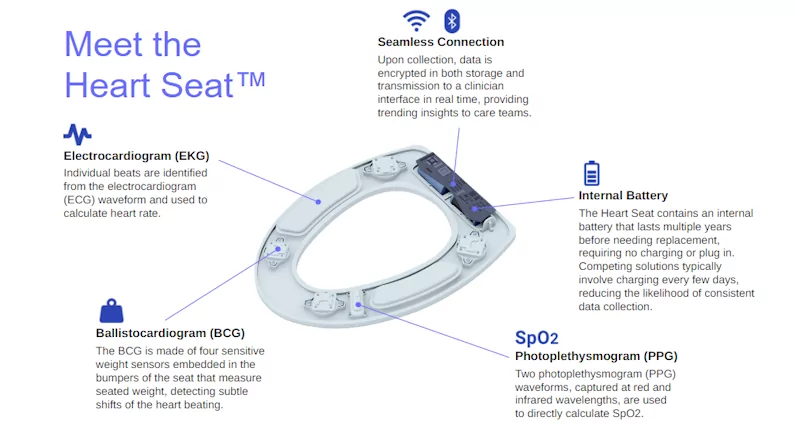
Figure 5—Features and sensors of the Heart Seat
The Heart Seat, a smart toilet seat for health monitoring, incorporates several key sensors to capture heart rate, blood oxygenation, and future clinical measurements, such as blood pressure. The four BCG sensors embedded in the seat’s bumper work in tandem with the ECG and PPG sensors to provide unique waveform reference points. By capturing information from multiple unique sources over the same period of time, the data gathered can help better define hemodynamic activity.
A comprehensive view of the electrical, mechanical, and peripheral activity of the heart removes the ambiguity that would otherwise be present from just one sensor. For instance, the electrical activity of the heart does not necessarily reflect the mechanical reality. Electrical impulses can propagate through the heart, but that does not mean the heart itself is constricting at that moment. Differences in the timing between these two data sources provide insights that might have otherwise been missed.
Reliable, periodic data collection assists in identifying trends in patient health. The Heart Seat was built to provide comprehensive data sets by addressing one of the most prominent issues in home monitoring: adherence. Minimizing required patient interaction reduces the risk of human error, and improves the likelihood of clinicians getting the data they need. While other sensor-embedded devices can struggle to achieve the necessary level of skin contact, the Heart Seat was designed to circumvent that challenge altogether. A toilet seat is naturally used multiple times a week without physician or nurse intervention.
As we look toward the future of healthcare, your expertise could be instrumental in unlocking the full potential of ballistocardiograms. If you share our vision and are interested in exploring potential partnerships, we invite you to connect with us, by reaching out to info@casanacare.com.
Recommended insights
Why Ballistocardiography is the Leader of Next Generation Cardiac Sensors
A century-old sensor carries the potential to revolutionize today's in-home care. The BCG's potential has been rediscovered.
Predicting the Unpredictable | How Managing the Supply Chain at a Startup is Full of Exciting Challenges
Interview with Shannon Clements, Supply Chain Manager at Casana
Remote Patient Monitoring Guide | What is RPM?
For most of human history, health care was delivered in or near the home. And while the movement toward centralized healthcare has provided great advancements in treatment and research, there is little doubt that something has...Read More
